Bacteriophage - Morphology, Life Cycle, Genetics and The Phageome (2/2)
Sep 30, 2020

Intro to Bacteriophage
In part 1, I touched on:
- What a bacteriophage is.
- The abundance of phage in the environment.
- The evolution of phage.
- The significance of phage in science and medicine.
- The history of their discovery.
Part 2: Phage Biology
In this section, we’ll cover:
- Morphology
- Replication cycles (and even how diet can influence them)
- The phageome in disease states
- Genetic composition
- Taxonomy
☕ Cup of tea at the ready — this one’s long!
Morphology
Phage are most famous for their icosahedral (20-faced) head/capsid, which houses their genetic material (DNA or RNA). The capsid connects to a tail with spidery, leg-like appendages.

The most abundant and studied group are tailed dsDNA phages, order Caudovirales.
In the human gut, there is about 1 virus-like particle (VLP) per bacterial cell, and most of these are dsDNA phages.
Tailed phages have three major parts:
- Capsid — where the genome is packed.
- Tail — the pipe through which DNA/RNA is ejected into the host.
- Adsorption apparatus — binding system that penetrates the host’s cell wall.
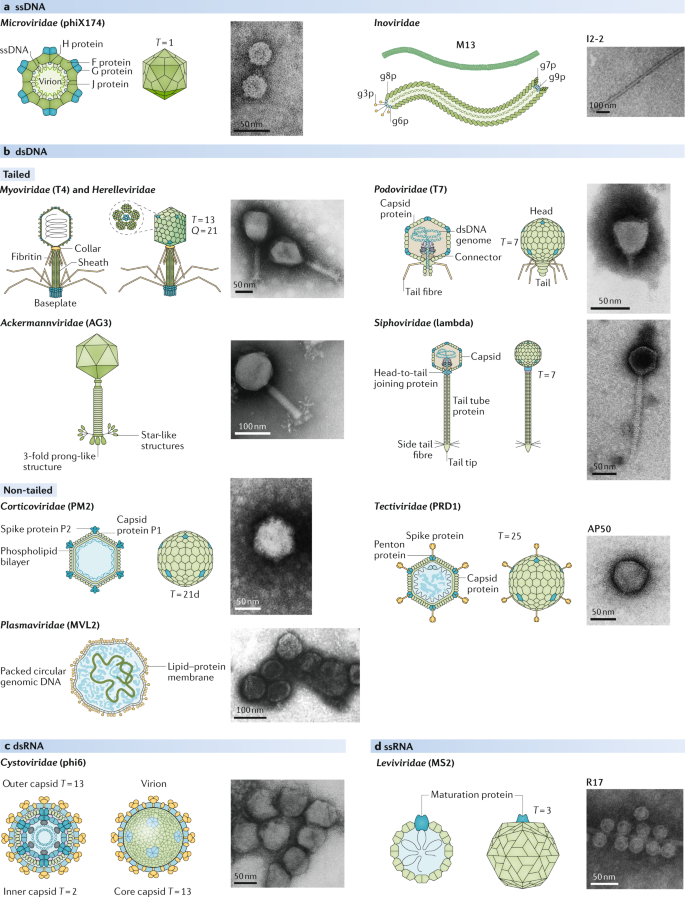
Phage diversity, genomics and phylogeny
Phage geometry is beautifully symmetrical, like molecular origami.
X-ray crystallography first resolved a phage capsid at 3.6 Å resolution (1 Å = 0.1 nm).
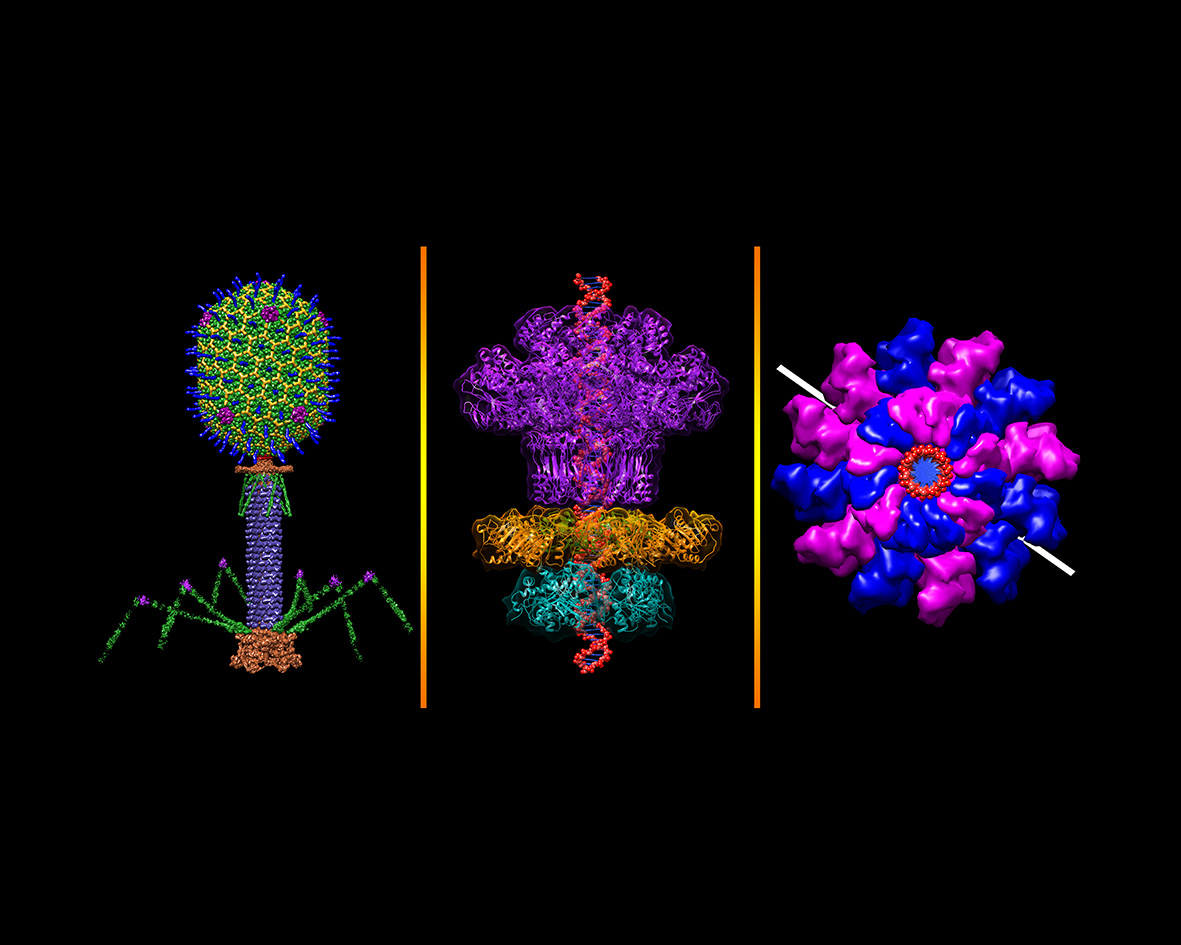
Credit: Dr Victor Padilla-Sanchez
Phage resemble a lunar lander — protein machines evolved for one task.
Their symmetry breaks at the head-to-tail interface (HTI), built around a portal protein valve that releases DNA.
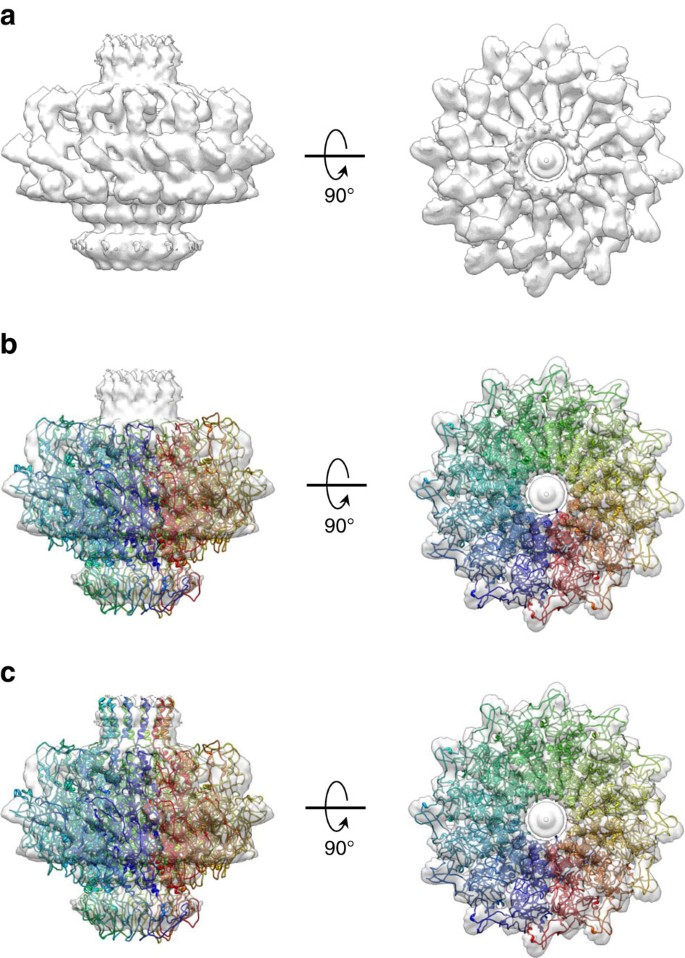
Architecture of the portal protein by cryo-EM
The tail acts like a syringe, injecting DNA into the host.

Bacteriophage organisation & function
💡 Recent work even explores giant viruses (e.g. Samba virus), which have conserved “stargate” portals — stable, star-shaped capsid openings that eerily resemble Alien movie pods.
Phage Life & Replication Cycles
Phages live a double life.
They can exist as:
- Lytic/virulent: infect, replicate, burst host → spread.
- Lysogenic/temperate: integrate DNA into host genome as a prophage, remaining dormant until conditions favor activation.

Some phage are strictly lytic (good for phage therapy), others toggle between states, and in the gut, additional states exist: pseudolysogeny, carrier state, chronic infection.
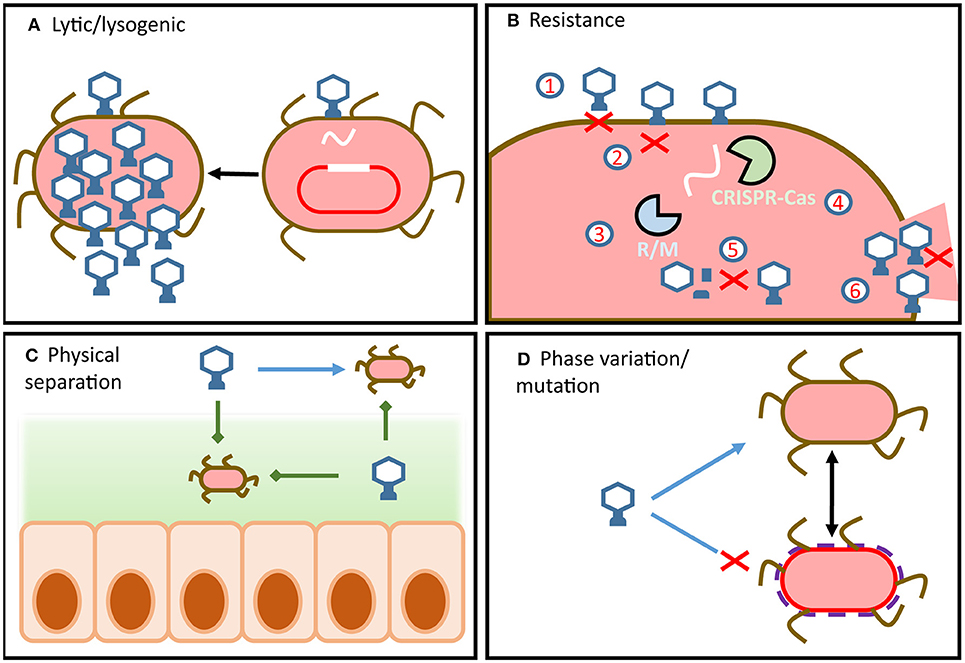
Gut bacteriophage: current understanding
Host–phage dynamics are complex: bacteria evolve defenses (like CRISPR), while phage evolve countermeasures.
Prophages
I’m fascinated by symbiogenesis — DNA becoming permanent residents in foreign cells. Prophages are a striking example.

Key points:
- ~50% of bacterial genomes carry prophages.
- Pathogens are more likely lysogenic.
- S. aureus carries 1–4 prophages; Salmonella species always carry some.
- Prophages disrupt genes, sometimes harm hosts, but also confer immunity, adaptive genes, and virulence factors.
Phage & Disease
The virome is now linked to disease.
In Crohn’s disease:
- The core virome disappears.
- Healthy guts: dominated by lytic phage.
- Diseased guts: dominated by lysogenic phage.

Inflammation may reduce bacterial populations → phage toggle to dormancy.
Food & Phage States
Phage cycles can be induced by chemicals. In the lab, mitomycin C forces prophages into lytic mode.
A study of 117 foods & additives found diet shifts phage cycles:
- Some foods induce lysis → more VLPs.
- Others repress phage → fewer VLPs.

Examples:
- Inducers: soy sauce, nicotine, sunscreen, antibiotics.
- Repressors: pomegranate (-89%), grapefruit extract (-89%), coffee (-49%), cinnamon (-88%), kombucha (-44%).
👉 Diet influences not just bacteria, but also phage dynamics.
Genetic Composition
Phages vary wildly in genome composition — each is unique.
Unlike vertical inheritance in eukaryotes, phage and bacteria thrive on horizontal gene transfer (HGT).
Methods of gene transfer:
- Transduction
- Gene transfer agents
Temperate phages are “gene brokers,” providing new genes for hosts and viral peers.
Phages act as gene-transfer particles, fueling bacterial evolution.
Transduction
- Generalized transduction: random bacterial DNA packaged into phage capsids.
- Specialized transduction: specific DNA near prophage site transferred.
This can spread antibiotic resistance genes.
A study showed phage help bacteria survive antibiotics by transducing resistance genes.
Even dormant prophages quietly spread genes, shaping microbial populations.

Genetic Mosaicism
Phages often have mosaic genomes: patchworks of DNA “modules” from many origins.
These modules encode key units for replication, assembly, and infection.
Horizontal gene swapping over billions of years produced extreme genomic diversity.
Taxonomy

Virus taxonomy is regulated by the ICTV.
The 2019 release lists 6,590 species.
Historically, phages were classified by morphology and genome type. Now, metagenomics is accepted in proposals.
But with rampant HGT, phylogenetics is messy. Instead, approaches like gene-sharing networks are emerging.

Bipartite gene-genome network of dsDNA viruses
Despite the challenge, identifying viral hallmark genes still anchors phage as bona fide viruses.
Closing thoughts
Phages are not just ancient predators, but evolutionary architects and biotech pioneers.
Exploring their genetics and physiology will be key to combating antimicrobial resistance and unlocking new biotechnologies.